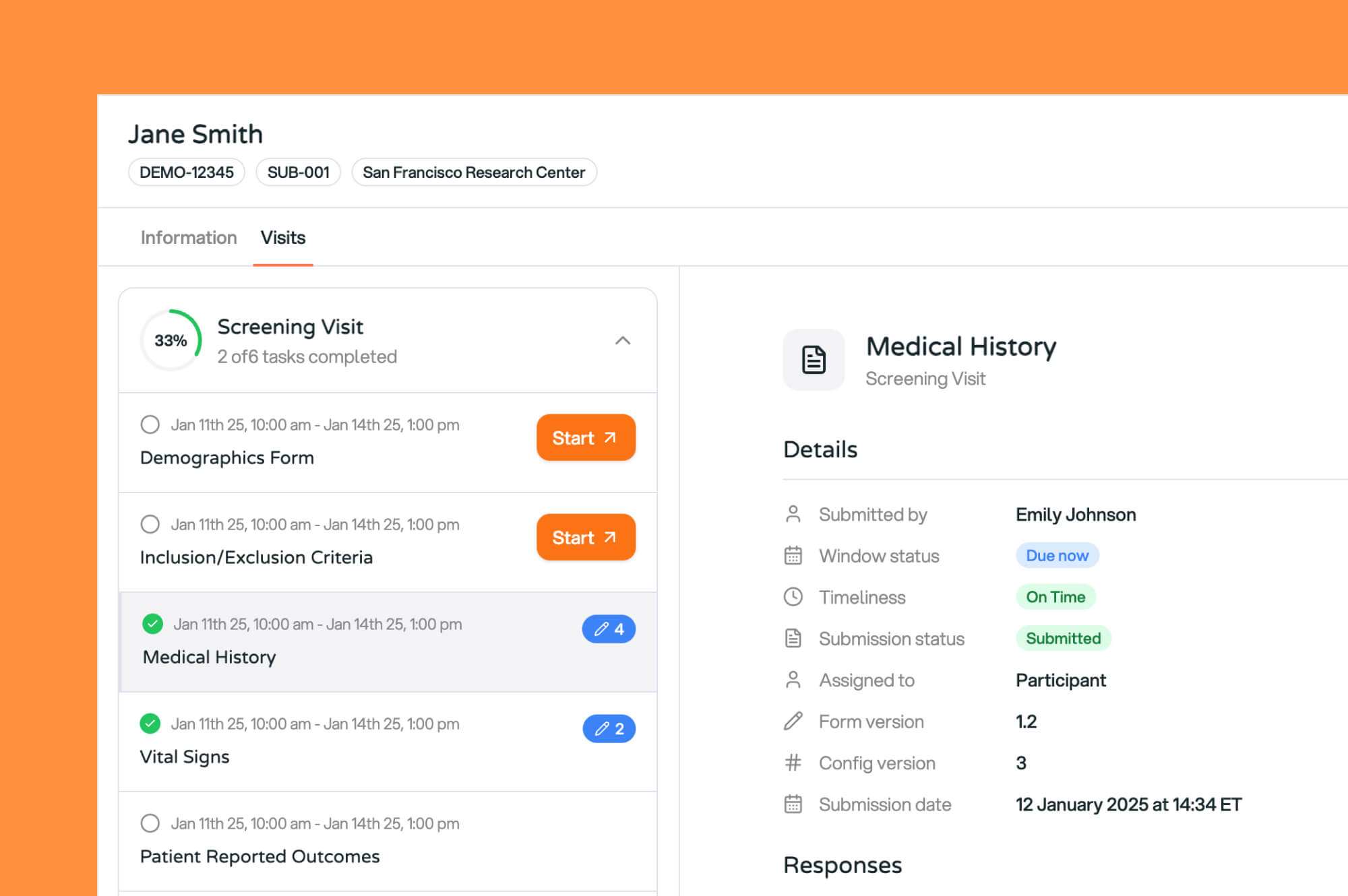
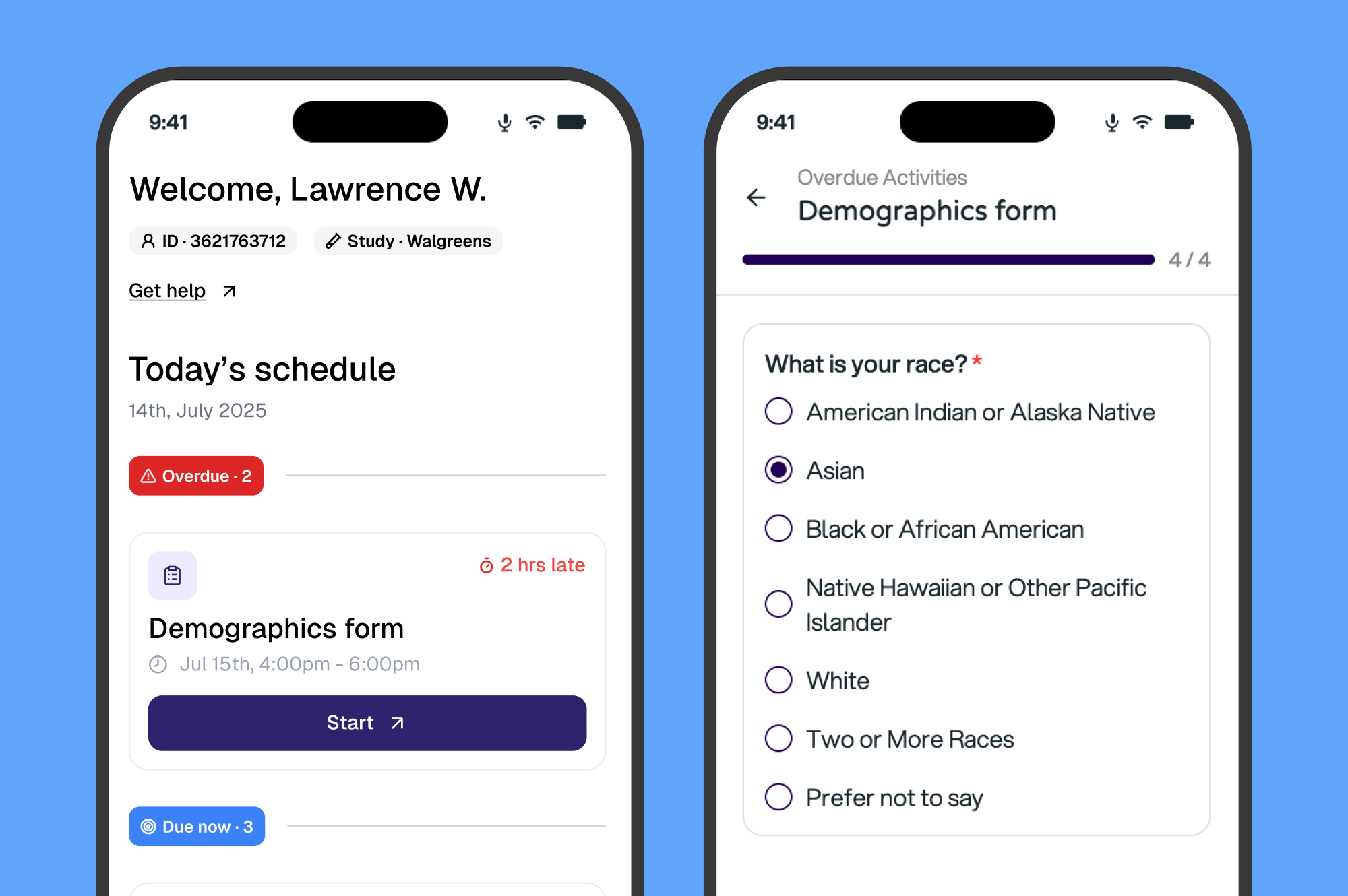
One eClinical Platform to Take You From Study Startup to Database Lock
Launch studies faster, capture cleaner data, and boost participant engagement, all on Curebase's AI-powered clinical trial technology platform.
Explore the Platform
Powering studies at leading organizations like:
















Building Modern eClinical Technology for Teams Like Yours
Whether you're a two-person biotech or a global CRO, Curebase adapts to how you work.
Our Impact in Numbers
0K+
Participants
0K+
Consents Signed
0.0M+
Data Points Collected
0+
Studies Powered
Interactive Demo
Try the Curebase eClinical Platform now, no sign up required.