Fully Integrated Electronic Data Capture
Curebase EDC is fully integrated into our platform, giving sites, sponsors, and CROs a seamless, user-friendly experience from ePRO to CRFs to query resolution.
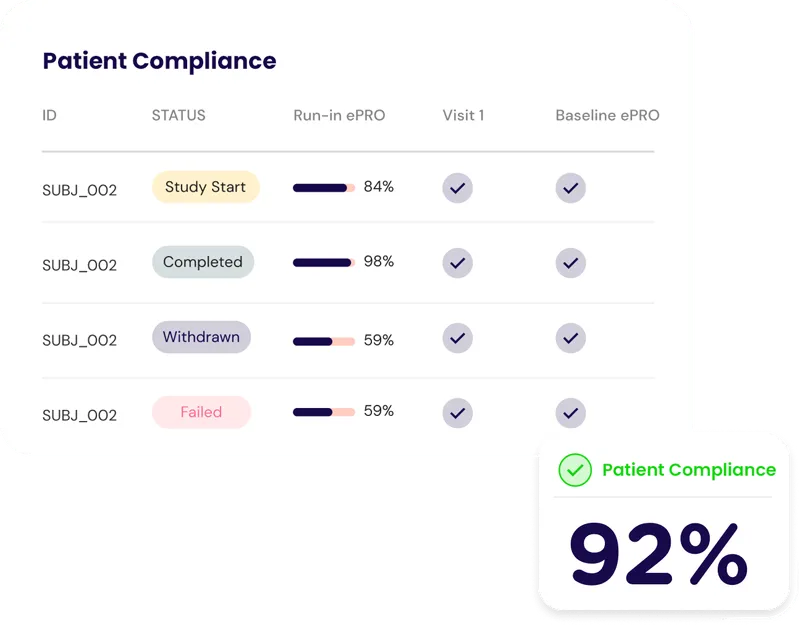
Smarter EDC for Seamless Clinical Data Management
All-in-One Platform
EDC fully integrated with ePRO, eConsent, and engagement tools for a structured workflow.
Streamlined CRF Entry
Elegant interfaces for case report forms, adverse events, medications, and log forms.
Powerful Data Review
Freeze, lock, and query with ease, built for monitors and site users alike.
Real-Time Insights
Dashboards and notifications keep everyone informed, from PIs to study teams.
Tablet-Ready Design
Optimized for site workflows with delightful experiences on both laptops and tablets.
Simple Setup
Configurable workflows and forms let you move fast without compromising quality.
EDC That Sites Actually Want to Use
Most legacy EDC systems were built for data managers, not for the coordinators and PIs entering data during a patient visit. Curebase Electronic Data Capture for Clinical Trials inverts that equation: clean forms, fast tablet input, real-time edit checks, and inline queries that resolve in the moment instead of weeks later.
Because EDC, ePRO, and eConsent share one data model, sponsors get a cleaner database with fewer reconciliations, and study teams stop juggling disconnected systems.
How It Works
Design
Configure CRFs, validation rules, and workflows with an intuitive form builder
Capture
Sites enter data on any device with real-time validation and edit checks
Review
Freeze, lock, query, and export CDISC-compliant data with full audit trails
Built for Sites and Sponsors
Curebase EDC pairs site-friendly data entry with sponsor-grade controls: role-based permissions, source data verification, freeze and lock workflows, and validated electronic signatures, all backed by a complete 21 CFR Part 11 audit trail.
Configurable edit checks catch issues at the point of entry, so monitors spend less time chasing queries and more time on meaningful data review.
Integrated With the Full Trial Stack
EDC connects directly with Curebase eConsent, ePRO, recruitment, and participant engagement on a single structured platform. ePRO data flows into the same database as CRFs, eliminating reconciliation work and giving study teams one source of truth from screening through database lock.
Why Curebase EDC Outperforms Legacy EDC
| Legacy EDC | Curebase EDC | |
|---|---|---|
| Setup time | Heavy build cycles | Configurable forms and reusable templates |
| ePRO and eConsent integration | Third-party connectors and re-keying | Same platform, shared data model |
| Query management | Siloed and asynchronous | In-platform, real-time, with site notifications |
| Tablet UX | Desktop-only, hard at the bedside | Native tablet and laptop experience |
| Data export | Custom mappings per study | CDISC-compliant exports out of the box |
